Understanding Entropy without Probability
In 1856, Rudolf Clausius coined the word entropy as a suitable name for what he had been calling the "transformational content of a body". The new word made it possible to state the second law of thermodynamics in the brief but alarming form: "The entropy of the universe tends toward a maximum." Thus, entropy is originally related to possible changes, not to chaos and probability as in subsequent work by Boltzmann and Gibbs.
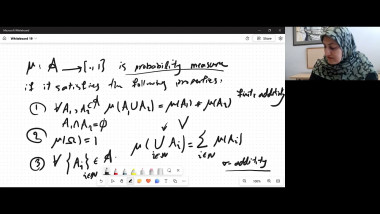
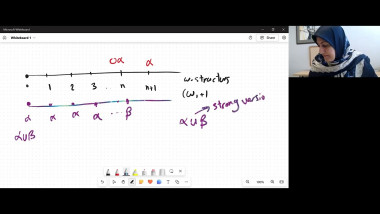
In the talk, I shall discuss a modern version of this view on entropy developed in joint work with Elliott Lieb. The key concept is a relation, called adiabatic accessibility, between pairs of states of macroscopic bodies. It will be explained how an essentially unique entropy emerges from a few simple properties of this relation.